DISCOVER neva vs
A DEFINITIVE SOLUTION for post aSAH Vasospasm
NEVA VS, SPECIFIC TO VASOSPASM TREATMENT
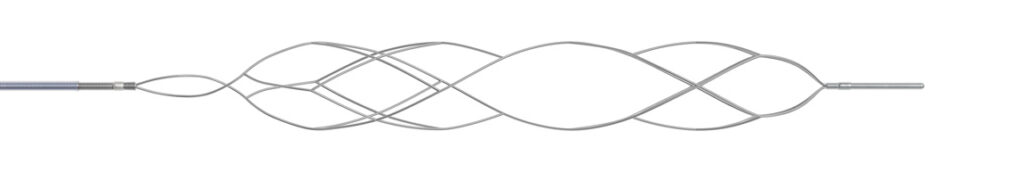
NeVa VS combines a smooth and continuous architecture and the deliverability of a stent retriever with an enhanced outward radial force to effectively dilate critically narrowed vessels.
CEREBRAL VASOSPASM
COMMON AND DEADLY
1 in 10,000 people experience an aneurysm rupture every year and up to 70% of these patients have to cope with vasospasm in the aftermath. This common complication of post-aSAH is also the leading cause if delayed morbidity and mortality.
NEW TREATMENT OPTIONS
TO REVERSE VASOSPASM
This life-threatening narrowing of brain vessels limits blood flow to the affected area, causing delayed cerebral ischemia and poor neurological outcomes.
Today, with advances in technology and new treatment pathways, cerebral vasospasm could be reversible.
Case images courtesy of Dr Arthur Grigorian, MD, Wellstar North Fulton Medical Center. Roswell, Georgia
NEVA VS FOR VASOSPASM: RECENT EVIDENCE
Click to watch the presentation highlighting NeVa VS as the game-changer in treating vasospasm after aneurysmal sub-arachnoid haemorrhage (aSAH) at the BRAIN CONFERENCE in London, December 2024.
“NeVa VS is a safe strategy to regain vessel diameter in severely narrowed proximal intracranial arteries”
– Dr. Ameer Hassan, Valley Baptist Neuroscience Institute, Harlingen, Texas
EXPANSION OPTIMIZED
FLOW MAINTAINEDNeVa VS is a patented, retrievable nitinol stent for post-aneurysmal cerebral vasospasm combining
smooth architecture, deliverability, and enhanced radial force to dilate critically narrowed vessels.
SPECIFIC
Designed For Vasospasm
Optimized Vessel Expansion
Distal Flow Maintained During Treatment
SUCCESSFUL
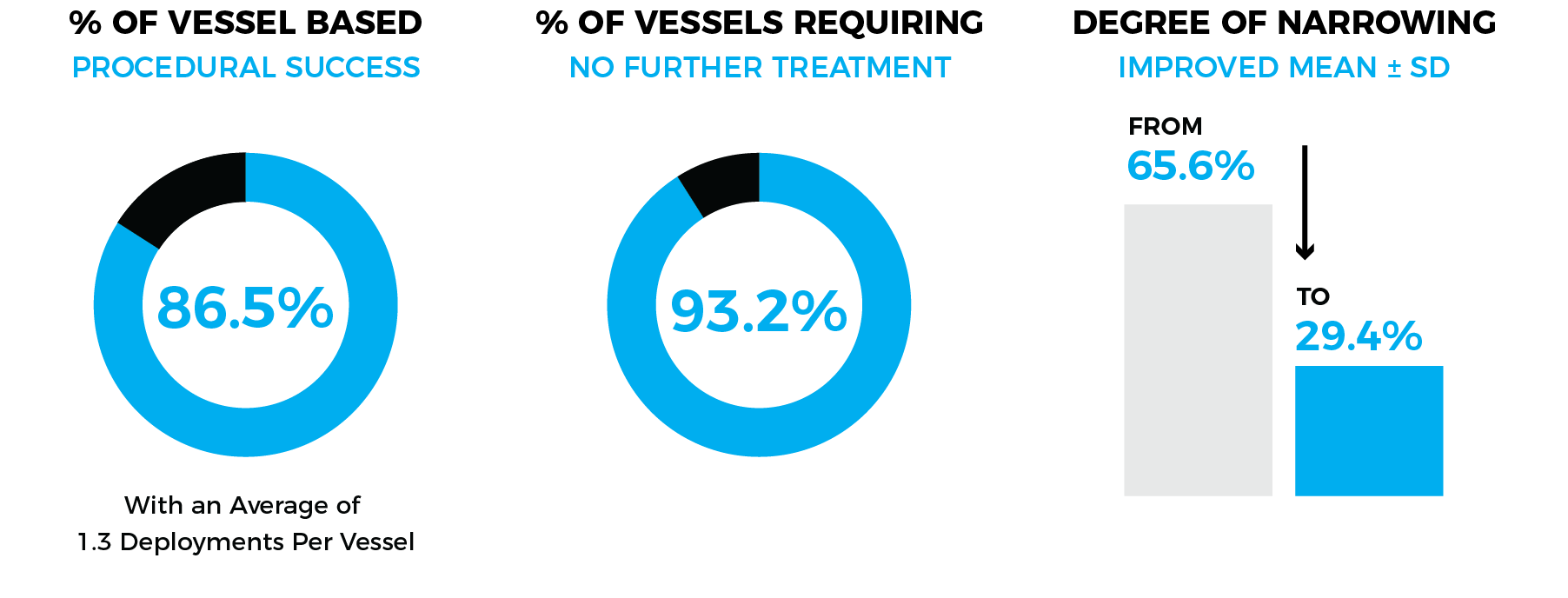
SAFE
No Vessel injuries or Ruptures
NEVA VS
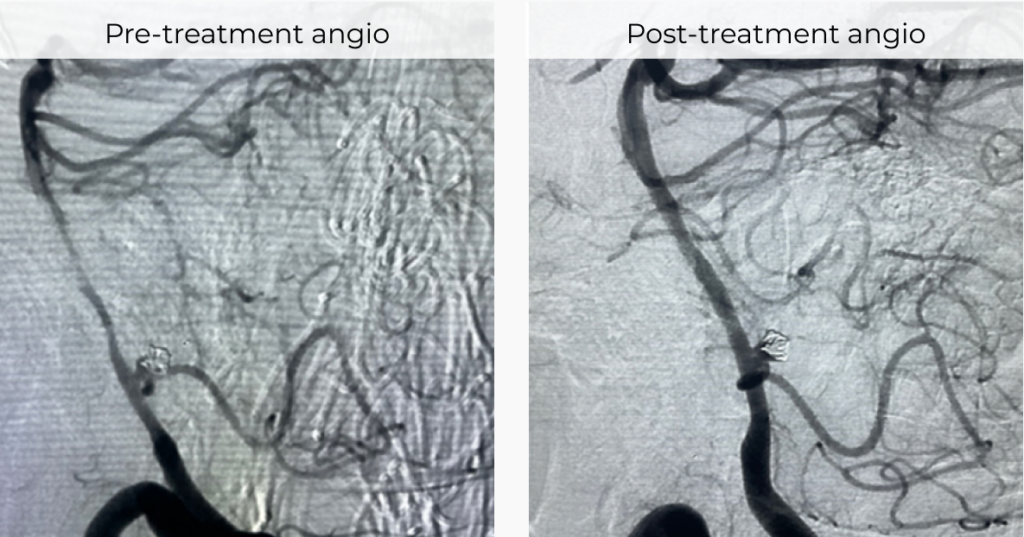
IN ACTIONMODERATE-SEVERE BASILAR ARTERY VASOSPASM SUCCESSFULLY RESOLVED WITH NEVA VS
Dr Ryan M Hebert, Yale School of Medicine, New Haven, Connecticut
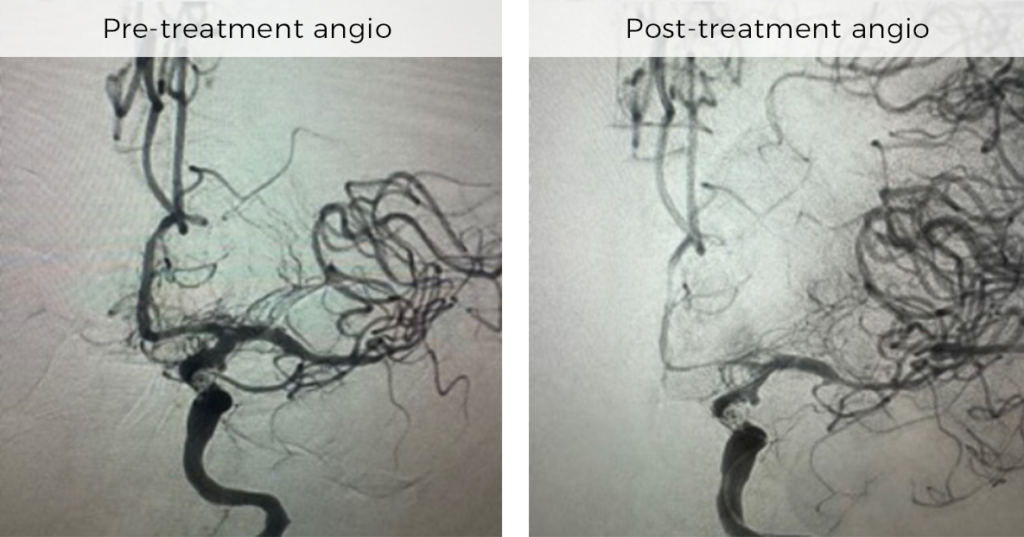
RIGHT MIDDLE CEREBRAL AND ANTERIOR CEREBRAL ARTERY VASOSPASM TREATED WITH NEVA VS
Dr Arthur Grigorian, Wellstar North Fulton Roswell, Georgia
FDA-APPROVED
AND CE-MARKED
NeVa VS is the first and only FDA HDE approved device for the adjunct treatment of symptomatic cerebral vasospasm following aneurysmal subarachnoid hemorrhage, and now also CE marked for vasospasm treatment.
Approved by FDA under HDE 210004. Note: The effectiveness of this device for this use has not been demonstrated”
A DEFINITIVE SOLUTION
FOR VASOSPASM
Would you like to know more?