ENGINEERED FOR
CORONARY THROMBECTOMY
enVast is the first and only CE-approved and FDA-cleared large thrombus burden retriever in patients with coronary occlusions.

enVast is the first and only CE-approved and FDA-cleared large thrombus burden retriever in patients with coronary occlusions.

Equipped with Drop Zone technology, the enVast coronary thrombectomy system has been proven to remove large thrombus burden (LTB) and create immediate reperfusion in 85% of cases. (n = 61)
(core-lab adjudicated)
72%
MBG 2-3 after enVast
85%
immediate reperfusion at enVast deployment
90%
TIMI 3
after enVast
98%
TIMI 3
end of PCI
Procedural Outcomes
0
1.6%
coronary embolization (all resolved)
Clinical Outcomes – 30 Days
0
3.3%
cardiovascular death (patients were in cardiogenic shock at admission)
designed to mitigate distal embolization and stroke risk
Vesalio is sponsoring the NATURE study, a randomized, multi-center clinical trial, comparing the safety and efficacy of enVast™ as an adjunct to conventional intervention versus the standard of care in MI patients with large thrombus burden.
clinicaltrials.gov ID NCT04969471
“enVast could provide a new therapeutic opportunity for myocardial infarct patients with large thrombus burden, who still represent a major challenge for procedural success and long-term outcomes.”
Prof Dr Marco Valgimigli
Istituto Cardiocentro Ticino, Lugano, Switzerland
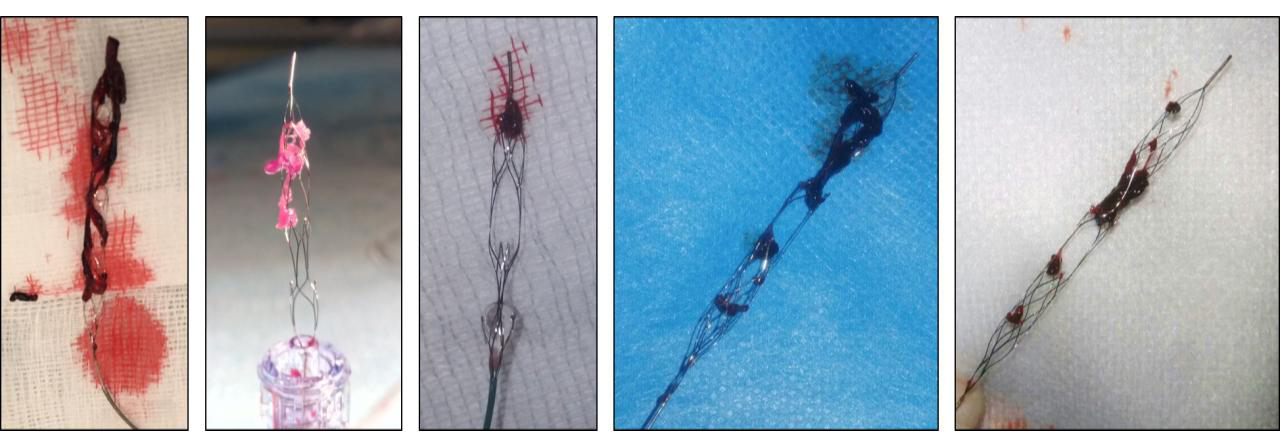
Precaution: The safety and effectiveness of this device for use in the treatment of ST-Elevation Myocardial Infarction (STEMI) have not been established. Complications from the use of this device in this manner could lead to death, permanent impairment, and/or the need for emergency medical intervention
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
by removing vascular occlusions and restoring blood flow
Please select your region to enter our world of innovation:
"*" indicates required fields