NeVa™ TO THE RESCUE
1st Pass Success with NeVaTM M1S (4.0 x 22 mm) after failure of 2 different devices
1st Pass Success with NeVaTM M1S (4.0 x 22 mm) after failure of 2 different devices
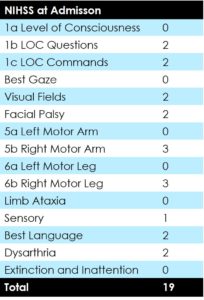
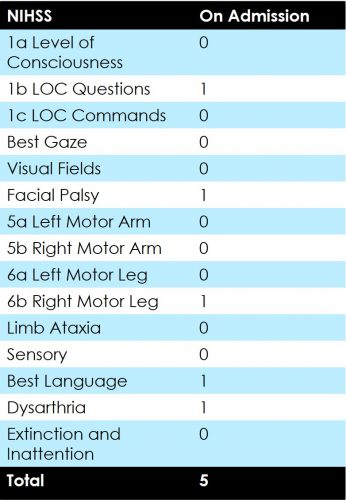
75 year old female patient presented with NIHSS of 19 presented 4 hours after symptom onset.
It was noted that the patient had had a knee implant surgery 3 days prior to her stroke. Additional risk factors included: Coronary Heart Failure, Hypertension, Diabetes, Atrial fibrillation
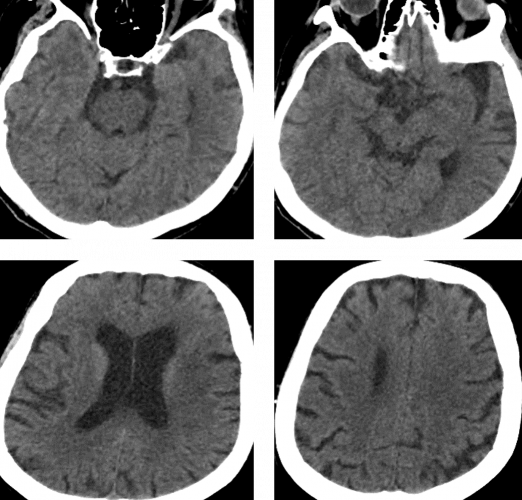
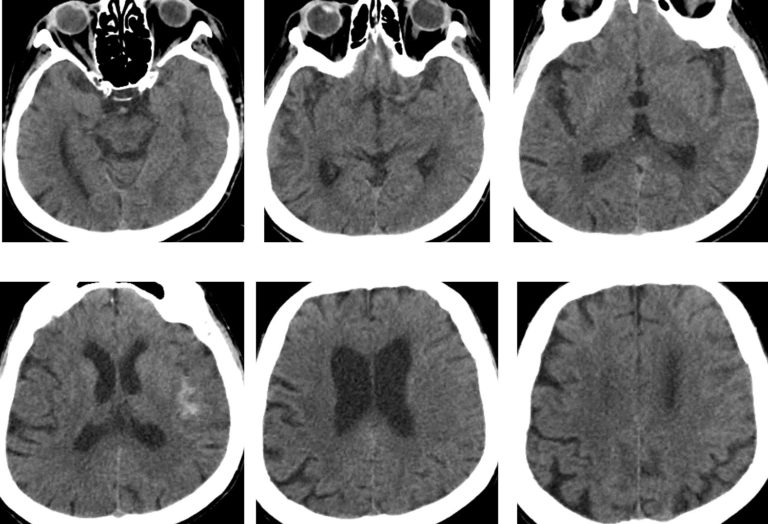
Imaging confirmed distal left MCA occlusion.

Professor Dr. Serdar Geyik
Istanbul Istinye University
Medical Park Hospital
Istanbul, Turkey
On admission CT ASPECTS was 10.
Professor Geyik and the Medical Park team decided to treat the patient with thrombectomy.
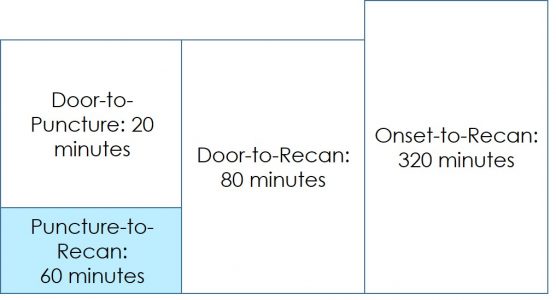
Since the occlusion was in a relatively distal branch, Dr Geyik initially tried two different brands of 3mm OD stent retriever devices. Following the failure of these two devices at achieving recanalization, he decided to give the shortest NeVa (M1S – 4 mm x 22 mm) a try. Full recanalization (TICI 3) was possible in a single NeVa pass.
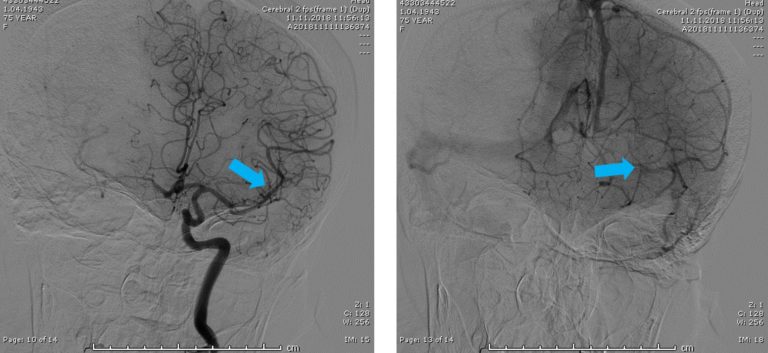
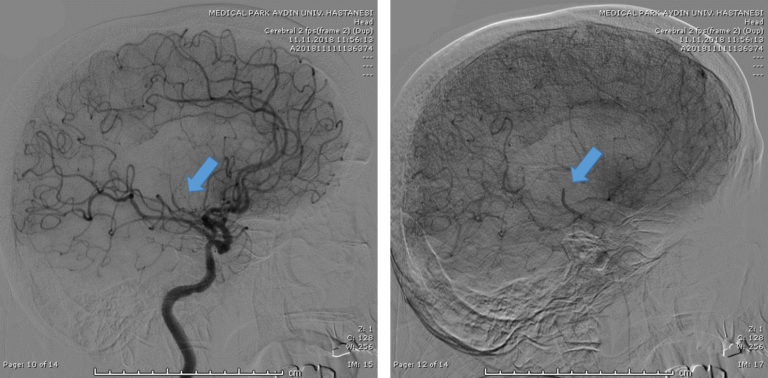
Access to the occluded site was possible despite extreme tortuosity.
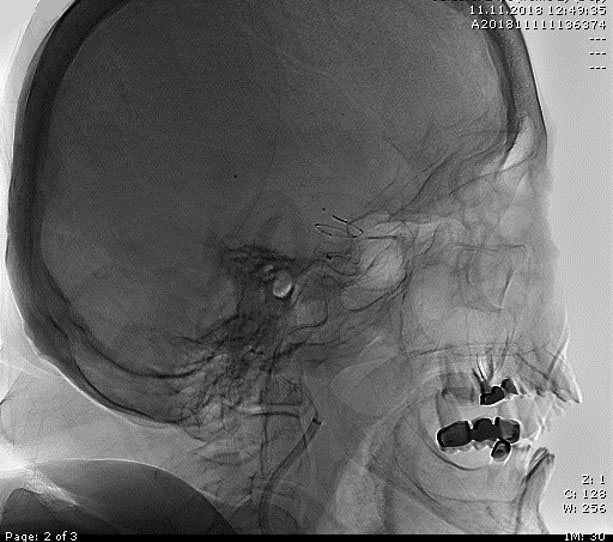
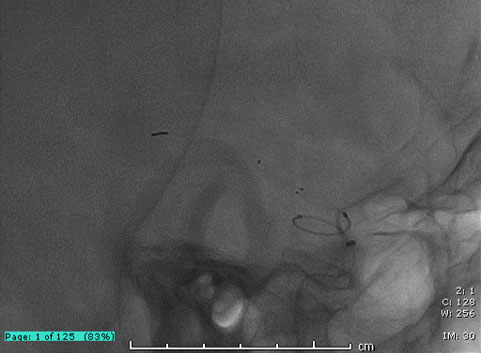
Note that the DAC was parked relatively proximally to the occlusion site due to the extreme tortuosity of the branch.
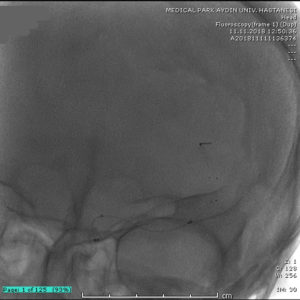
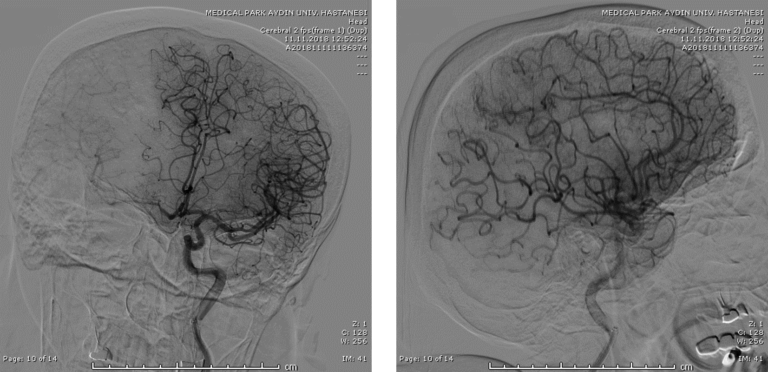

24 hour NIHSS of the patient was 5.
At VESALIO, we feel blessed to be part of the stroke field where together with these dedicated physicians, we can make an incredible impact on people’s lives.
Thank you Istanbul!
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
by removing vascular occlusions and restoring blood flow
Please select your region to enter our world of innovation:
"*" indicates required fields